Copyright © 2014: Manila Retina Specialists All Rights Reserved
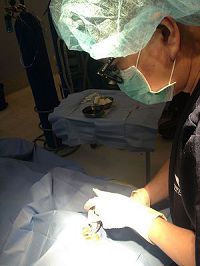 Fig 2. Dr. Uy injecting anti-VEGF in a patient with Diabetic Macular Edema. Sterile injection technique in an operating room minimizes risk of infection.
Fig 2. Dr. Uy injecting anti-VEGF in a patient with Diabetic Macular Edema. Sterile injection technique in an operating room minimizes risk of infection.
This is a short procedure involving placing a tiny amount of drug, containing antibodies inside the vitreous cavity. Numbing (anesthetic) drops are placed in your eyes. Antiseptic drops are also given to disinfect the eye.
The patient is placed in bed and the eye is held open with lid speculum. The eye is numbed with lidocaine (anesthetic) after which the injection is done. Artificial tear drops are placed in the eye and no patch is needed. The entire procedure lasts around 10 minutes.
The patient is instructed to return in a week or so, but sooner if there is blurring of vision or severe pain. You can usually perform work with no restrictions. You may see a floater, which is due to the drug. A little redness around the injection site is common, which we call subconjunctival hemorrhage.
There is a small chance of heart attack or stroke, but this maybe due to patient's age and comorbidities, and not necessarily caused by the drug. Infection is rare, but may occur in 1 per 7000 injections. Some people may develop allergic reactions may also occur, so its important to alert your physician especially if you develop a lot of floaters, or blurring of vision after the injection.
Five brand names are used, Lucentis (Ranibizumab), Avastin (Bevacizumab), Eylea (Aflibercept), Brolucizumab (Vsiqq), or Vabysmo (Faricimab). Various trials have compared the first two, including the CATT trial. The studies show that they are virtually of the same efficacy, for AMD (macular degeneration), but Lucentis is safer in terms of almost having little chance of contamination, because of the way it is packaged. Due to affordability issues, Avastin is more widely used, though we have to remind patients that this is off label. Aflibercept has very good results for diabetic macular edema, and also for Polypoidal Choroidal Vasculopathy (PCV). Faricimab is the latest addition to our available medicines, which targets additional receptors called Ang-2.
Ozurdex is a steroid implant that is injected into the eye. Its main advantage is its delivery system, in the form of an implant. Its duration of action is 3-4 months, unlike the anti-VEGF which lasts anywhere from 4-16 weeks only. Its main use is for retinal vein occlusions, and macular edema. Its side effect is development of cataract and increased eye pressure, so we use it in patients who already had or will soon undergo cataract surgery. Eye pressure is checked every 6 weeks, and drops maybe prescribed to lower the pressure.
Off label means the drug was manufactured and US FDA approved for a different purpose, eg Avastin is used for colon cancer. It does not mean its bad, its just that the main indication is different. In real world, off label drugs have always been used, eg aspirin is now used for clot prevention, but it was approved for fever and pain.
Treatment may consist of initial 3-5 loading monthly doses, and some may need maintenance phase depending on the response of the patient.
Consult your Eye MD or Retina Physician as to which is best for your condition.
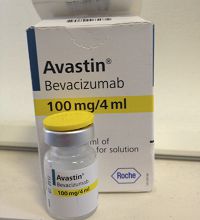 Fig 3 Avastin vial for off label injection of anti-VEGF. Fig 3 Avastin vial for off label injection of anti-VEGF. | 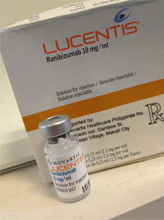 Fig 4 Lucentis vial is FDA approved for eye injection. Fig 4 Lucentis vial is FDA approved for eye injection. |
